
Prompt Images
In fact, the mere act of opening the box will determine the state of the cat, although in this case there were three determinate states the cat could be in: these being Alive, Dead, and Bloody Furious.
― Terry Pratchett, Lords and Ladies
1. A Series of Unfortunate Experiments
In a piece last year I wrote that black holes are really, really strange things. And it’s true, they are. But, to be honest, the strangest features of relativity (of which black holes are one) look down right pedestrian when compared to the all-consuming-bizarro-ness that is quantum physics.1
Relativity was conceived, essentially, in Einstein’s mind over the course of a few years while he passed as a so-so Swiss patent clerk and demonstrated arguably the greatest reason to support a generous government bureaucracy. Quantum physics was what physicists were able to scrape off the lab floor after a series of unfortunate experiments conducted over the first few decades of the 20th century.
Consider the Double Slit experiment, the archetype for experiments that show the bizarre things that can happen in quantum physics. Things that shouldn’t be possible.
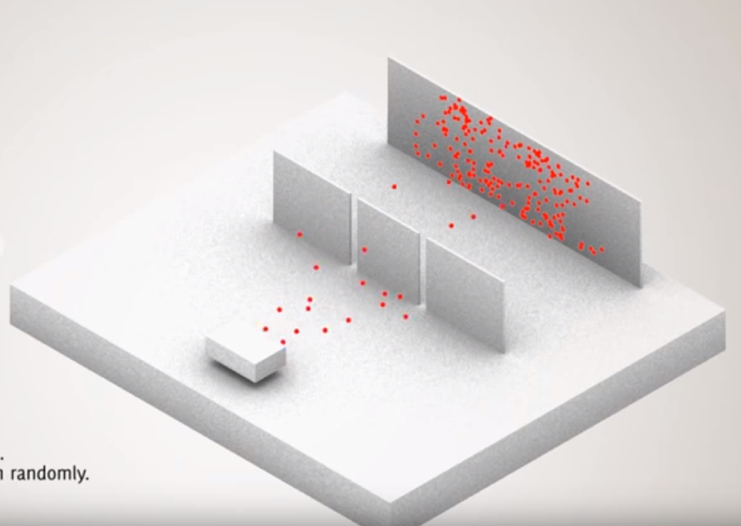
Here’s the gist of the experiment: you have a device (an electron beam gun) that fires out individual electrons at a screen. You can’t see individual electrons with your eyes so the screen is setup to “detect” where they land. In between the electron beam gun and the screen you put a wall with two “slits” (thus, the Double Slit experiment).

Double Slit experiment Setup: All images are courtesy (i.e., screen grabs) of this nifty YouTube video produced by www.toutestquantique.fr — the video is less than two minutes and well worth a watch.
Now the experiment goes as follows: aim the electron gun towards the two slits and fire away. The “aim” of the electron gun isn’t precise, so some will go through one slit and some through another, and all will eventually find their way to some particular spot on the screen. If you keep firing the gun you will get a bunch of spots where various electrons hit the screen (shown as orange dots in the images below).
OK, so if you haven’t seen this before, here’s a question: what pattern to do you expect to see on the screen after the gun has fired many electrons at the screen?
Well, if electrons behave like little tiny balls (e.g., BB’s), you wouldn’t expect to see much of a pattern on the other side. Each electron would go through one slit or the other, and then, depending on the direction it’s traveling in, it would hit some (random) location on the screen. If you did this a bunch of times the “pattern” might look like this:
See the interesting pattern? No, right, because there isn’t one.
It turns out, though, that electrons don’t actually behave like little BB’s. Because when you run the experiment here’s what you get:

Wait, what? Why are the electrons acting like a bunch of cliquish high schoolers?
Unlike the BB’s, the electrons do pile up in an interesting pattern on the screen. They almost exclusively show up in “bright” bands on the wall, separated by “dark” bands. These dark bands are some kind of quantum no man’s land where the electrons aren’t allowed to go.
Also, I should mention that if you cover up one of the slits, the pattern goes away.
If this whole thing doesn’t weird you out a bit, I promise it is only because you haven’t thought about it hard enough.
Maybe you believe there’s some underlying “classical” explanation. Maybe it has to do with how the electrons bounce off the interior walls of the slit as it passes through, or motions of air molecules, or some microscopic detail we haven’t considered.2 But how could any such explanation depend on there being one or two slits? An individual electron (allegedly) goes through one or the other slit. How then can said electron’s behavior be affected by the presence (or lack) of the slit it doesn’t go through?
Here’s how Richard Feynman, one of the greatest physicists of the last century, said about this experiment in a set of lectures he gave to a bunch of super nerds at CalTech:
We choose to examine a phenomenon which is impossible, absolutely impossible, to explain in any classical way, and which has in it the heart of quantum mechanics. In reality, it contains the only mystery. We cannot make the mystery go away by “explaining” how it works. We will just tell you how it works. In telling you how it works we will have told you about the basic peculiarities of all quantum mechanics.
So, let’s be clear, I’m going to try to give you a new way to think about quantum mechanics, but ain’t nobody walking away from this piece feeling like Oh, I totally get it now. The mystery is solved!
2. Wave-Particle Duality My Ass
The Double Slit experiment is taught in every quantum mechanics textbook ever written. Physicists basically wet themselves in their unbridled excitement to speak about this phenomena to anyone who will listen.3 The predominant pedagogical purpose of going through the experiment is to demonstrate so called wave-particle duality. That is, the idea that particles such as electrons sometimes behave like waves and sometimes behave like particles.
How does this experiment demonstrate that electrons can behave like waves? Well, waves can “interfere” with one another to create a so-called diffraction pattern, which looks exactly like the bands we see in the Double Slit experiment. In fact, the Double Slit experiment was originally an experiment done with light, way back before anyone started spouting mumbo jumbo about light being made of “photons” or electrons being made of “waves.” The same interference pattern can also be seen in water waves. Interference is a wave phenomena, not a classical or quantum phenomena.

This guy gets wave interference! Whenever you see two different circular ripples in a body of water “interact” to create a new pattern — that’s interference. If there’s a place where the peak of one wave is equal and opposite to the trough of another wave they cancel and you get breaks in the otherwise circular patterns. (Photo credit)
Here’s the thing, though. The wave-particle duality explanation has never sat well with me. It isn’t that I think the description is wrong. I just think it’s hella confusing. First, for non-physicists it’s a confusing way to explain the Double Slit experiment, because most people don’t know much about the physics of waves. This means you first have to explain how waves work before you can explain quantum physics. But my bigger gripe with this “explanation” is that the “electron wave” that arises from this way of thinking isn’t a physical wave in the usual sense — it’s a wave of probability amplitudes (we’ll get to those soon). And so when you say the electron is like a particle and like a wave, the “like” is doing a hell of a lot of work there.
3. Is There A Better Way?
Our question is then, is there a better way to try and understand what is happening in the Double Slit experiment and where the quantum weirdness comes in?
Yes, I think so. Einstein stumbled upon relativity when he questioned the Newtonian conception of space and time. It turns out you can do something sort of similar to arrive at quantum mechanics. The difference being that instead of questioning the nature of space and time we’ll be questioning the nature of – and you may want to take a seat before I finish my sentence here – probability itself.
But look at that… our time is up. Guess you’ll have to wait to hear more about this in Part 2.
Footnotes:
1 Fun fact: Einstein invented the theory of relativity more or less by himself, but he also helped kickstart the whole quantum physics thing. The birth of quantum theory is often said to begin with work done by Max Planck in 1900. In his work, Planck realized that he could fix a problem that had been giving physicists fits by assuming that energy inside a particular system was “quantized,” that it could only exist in multiples of some number. But, at the time, nobody thought this “quantized energy” was an intrinsic property of the physical world, only some emergent property of this particular system. It was Einstein who made the bolder claim that light was itself made of little “quanta” in a 1905 paper in which he gave an explanation for the strange experimental result known as the “photoelectric effect.” It’s this idea that won him his nobel prize in 1921, not his ideas about spacetime and gravity.
2 Let’s put aside the fact that versions of this experiment have been done for over a hundred years and no one has yet to provide a plausible “classical” explanation.
3 As do people who write popular science explainers, apparently.


