
Prompt Images
In Part I, we explored the likelihood of future nuclear explosions. It appears that the answer is, to the delight of Xanax manufacturers, somewhat likely. Today, let’s consider the source of power in these destructive weapons.
1. Energy and Power
To understand how we get to civilization-ending explosions, let’s review the basic ideas behind energy and power sources.
The most fundamental concept in physics is this: Energy is never created or destroyed— it just gets passed around. At some fundamental level, all that is ever happening in the world is energy changing hands between different objects —be those particles or people. In a way, physicists are nothing more than nature’s accountants tracking the flow of funds.
When a cue ball smacks into a 9 ball, the cue ball just hands over all its kinetic energy to the 9 ball. But in some cases, energy can be transformed: gravitational energy to kinetic energy (i.e., falling), chemical energy to electrical energy—which is the very process that powers your iPhone, or, alternatively, causes your Samsung phone to burst into flames.
Another really important thing to know about energy is this:
Everything in the universe, be it balls, atoms, or Kardashians, is constantly looking to move to a state of lower energy.
A ball that rolls down a hill and stops has moved from a higher (literally, higher up the hill) energy state to a lower energy state. As the ball is rolling down the hill it experiences friction with the hill and loses some of its energy as heat (in the parlance of physics 101, some of the ball’s gravitational potential energy has been converted to heat energy).
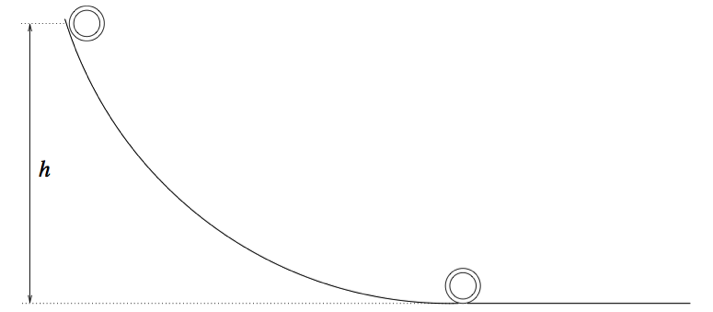
You can use these facts about energy (that it is never destroyed and that all systems are looking to move to a lower energy state, if possible) to create a “power source.” A power source is simply a system X that is not yet in its lowest possible energy state. To get the power out, you have to somehow “trigger” the system to move to a lower energy state, thereby releasing excess energy in the process. This excess energy is used to power some other system Y, be it a car or an iPad.
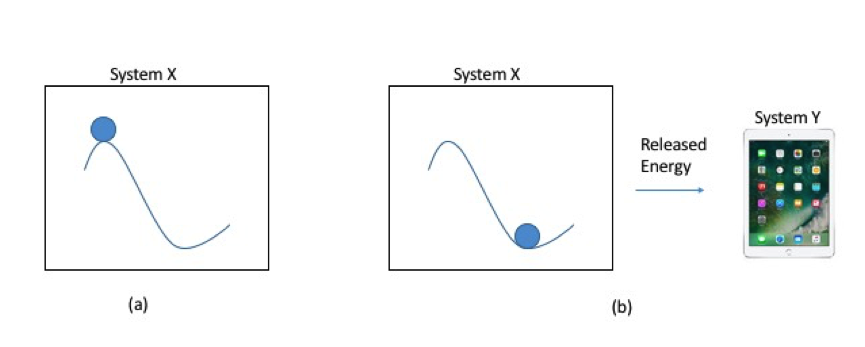
Power Source: (a) System X is not in its lowest energy state. (b) System X drops to a lower energy state and releases the excess energy that can be used to power System Y.
If System X releases the energy really, really quickly, you no longer have a very useful power source. You have a bomb.
2. Atomic Energy
The key to unleashing atomic energy is to recognize that an atom is, like a ball sitting atop a hill, just another system that isn’t yet in its lowest energy state. Meaning you can tap into its power. You just need to understand how to coax the atom into lowering its energy state and releasing the excess energy.
Now there are, in fact, a couple of ways that an atom can change its energy state. One is to hook up with other atoms to form molecules. Atoms like to team up because they can often share electrons, which reduces each atom’s individual energy state. When they do this, they form a chemical bond.
The energy released when atoms form these kinds of bonds is the source of energy we extract from burning fossil fuels. It’s also the source of the explosive power behind dynamite, TNT, and nitroglycerin.
There’s another way that atoms can change their energy, though. They can fuse together or break apart to form not just molecules, but completely new atoms. This is very exciting stuff, not just for science nerds, but also for sourcing energy.
Recall that an atom is made up of a nucleus surrounded by one or more electrons. That nucleus is a little ball further made up of protons and neutrons. It’s these nuclei that actually fuse together (fusion) or break apart (fission) in a nuclear reaction.
And when nuclei fuse or break up, they may also release energy.
If you stop and think about it, an atom with more than one proton (i.e., any atom other than hydrogen) shouldn’t exist. Protons are positively charged particles, and we all remember from Physics 101 and our refrigerator magnets that opposites attract, and like-charges repel. The repulsion felt by protons in the nucleus should be enough to send them flying apart. Right?
The reason protons are able to coexist in the nucleus is that they are bonded (along with neutrons also in the nucleus) by the strong nuclear force. This is a decidedly different force from the electromagnetic force that is behind chemical bonds between molecules, and it only operates inside an atom’s nucleus, because it dies off very quickly with distance. As its name belies, it is a very strong force, vastly stronger than the electromagnetic force.
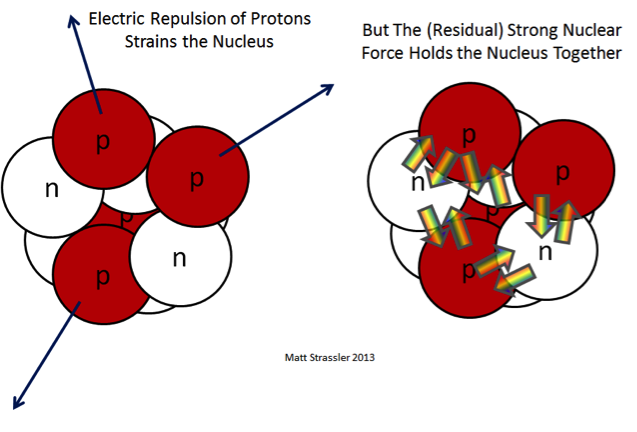
In the nucleus, then, you have the strong force operating to hold protons together. However, the electromagnetic force is still present, even if it’s not nearly as powerful as the strong force over short distances. The energy state of the atom is dependent on both of these forces.
As it turns out, with atoms there is an ideal atomic state that corresponds to the number of protons (and neutrons) present that best “balances” the effects of these forces and allows the atom to settle into its lowest energy state.
That ideal state is iron.
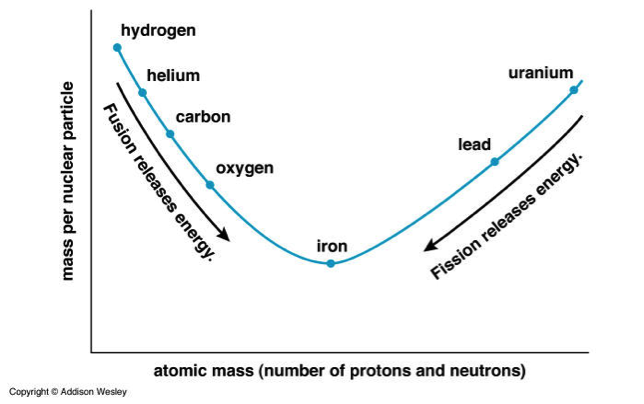
And the key to understanding nuclear reactions is this: all atoms want to be more like Iron, man.

And this is the core of how nuclear power works. Bond smaller elements (fusion) or break apart bigger elements (fission), so long as the result is one or more atoms closer in size to Iron, that platonic ideal of atomic structure, and you will release energy. And because the strong force is at play, the energy stakes are higher, you will get a much greater bang for your buck than from a chemical reaction. Pun intended.
Of course you don’t just get a mushroom cloud from splitting a single atom. The key is to start a self-sustaining chain reaction where one atom splits, and pieces of that splitting atom collide with a nearby atom and cause it to split, and so on. In 1942 the U.S. created the Manhattan Project to put this theory of nuclear explosions (which had been figured out years before) into practice. It only took 130,000 people, $2 billion, and dozens of the smartest people on Earth to make it happen.
3. Anatomy of Nuclear Blast
There are different kinds of nuclear weapons. In an atomic bomb (or A-bomb), the type of nuclear bomb developed (and dropped) during World War II, you start with a heavy radioactive element like plutonium 239 and trigger it to undergo the aforementioned chain reaction. This chain reaction happens in about 0.00000008 seconds—or about the amount of time the current commander-in-chief spends reading his weekly intelligence report.
During that chain reaction, a boatload of energy is released from the splitting plutonium atoms. This energy heats up any surrounding material to a balmy 10 million degrees. If you are wondering—how hot is 10 million degrees?—consider that the surface of the sun is only about 5,000 degrees. Oh, so it’s only 10,000 times hotter than the surface of the sun. Cool.
At 10 million degrees, you can do some damage. First, you can vaporize everything around you —plutonium, the bomb casing material, dirt, air. It all gets eviscerated into individual atoms, some of which have been stripped of their electrons.
These atoms are in really, really energetic states. Some of this energy is immediately released as X-rays and other thermal radiation. Most of the rest of the energy is released as shock-waves in the air. The result is an expanding fireball:

…and an expanding wave of sudden pressure changes that can do this to a house:

Oh, and if the fireball and the house destroying shockwave doesn’t get you, there’s always the radioactive fallout to finish you off more slowly.
So how far away do you need to be to avoid the fireball and/or shockwave?
Well, back when our parents were kids, you could survive a nuclear explosion if you were a mere mile or two away. But thanks to the Cold War, we were able to create much more destructive bombs, like the hydrogen bomb.
Here’s a nice comparison between the damage done by your grandmother’s bomb and a more modern nuclear bomb:
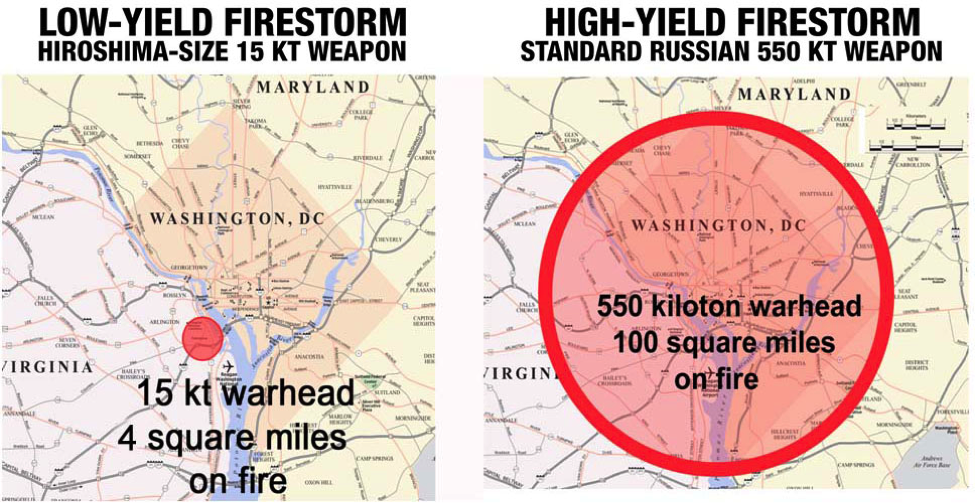
Image Credit: http://www.nucleardarkness.org/nuclear/highvslowyield/
The image on the right really gives new meaning to “inside the beltway.”
The thing is, a 550 kiloton bomb isn’t even close to the biggest nuclear bomb that’s ever been detonated. In 1961 the Russians pulled the old “That’s Not a Bomb, This is a Bomb,” move and tested the 50,000 kiloton Tsar Bomba. During that nuclear test, all buildings in the town of Severny, located 34 miles away from the blast site, were destroyed. The heat from the explosion was strong enough to induce third degree burns as far as 60 miles from the blast site. To be clear, that means a Tsar Bomba dropped on DC could cause third degree burns to folks in Baltimore, which is only about 40 miles away.
Purportedly, the Tsar Bomba was actually “dumbed down” for the nuclear test and could have reached the 100,000 kiloton mark had the engineers fully tricked it out. And there’s really no limit to the explosive power of a nuclear weapon, so long as you keep adding additional nuclear material.
4. It Could Be Worse
So to summarize what’s keeping me up at night, it’s easy to imagine a nuclear weapon detonating, perhaps by accident, at some point in the future. And that would be catastrophic because nuclear weapons are arguably the most destructive things man has created to date.
But it could be much worse! Because nuclear weapons aren’t even the most explosive energy source that we know about. An antimatter weapon, for example, would release a much greater amount of energy per mass that a nuclear weapon. But that’s a topic for a separate post and/or Dan Brown novel.
Also, to be totally clear, no nuke in existence could cause as much damage as a decently sized asteroid colliding with the earth. And we are apparently overdue for one of those.
If this is all a bit overwhelming, I understand. There’s not much we can do about asteroids or yet-to-be-created antimatter weapons. Even on the nuclear front there’s little we can do at this point—it’s Rick Perry’s problem now.
What I can suggest is the following: Find a good meditation app. Hold your loved ones close. And, of course,



